JVC to launch the CL-R813, a 32-inch medical imaging monitor
8-megapixel wide color monitor for displaying various diagnostic images on one workstation
Jun 30, 2022
JVC will release the CL-R813 (32-inch 8-megapixel color LCD monitor) as of medical image display monitor (hereinafter referred to as "medical monitor") in July 2022.
| Item name | Model name | Recommended retail price (tax excluded) | Release date |
|---|---|---|---|
| 32-inch 8-megapixel color LCD monitor | CL-R813 | Open price | July 2022 |
Project’s intention
In recent years, the evolution of medical equipment has made it possible to handle a large amount of image data. More and more doctors are using multiple medical monitors side by side, putting a burden on imaging diagnosis and requiring larger working space.
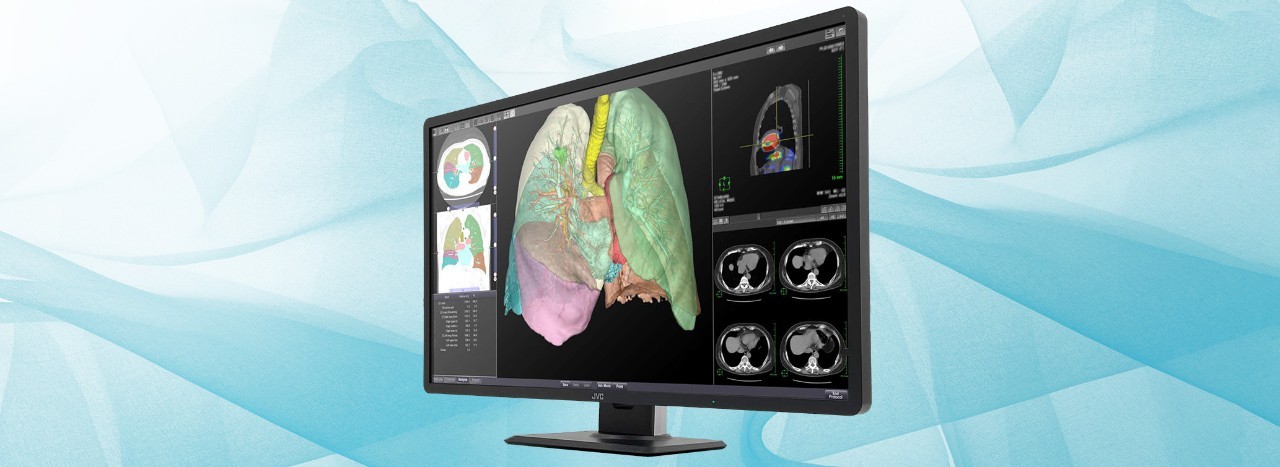
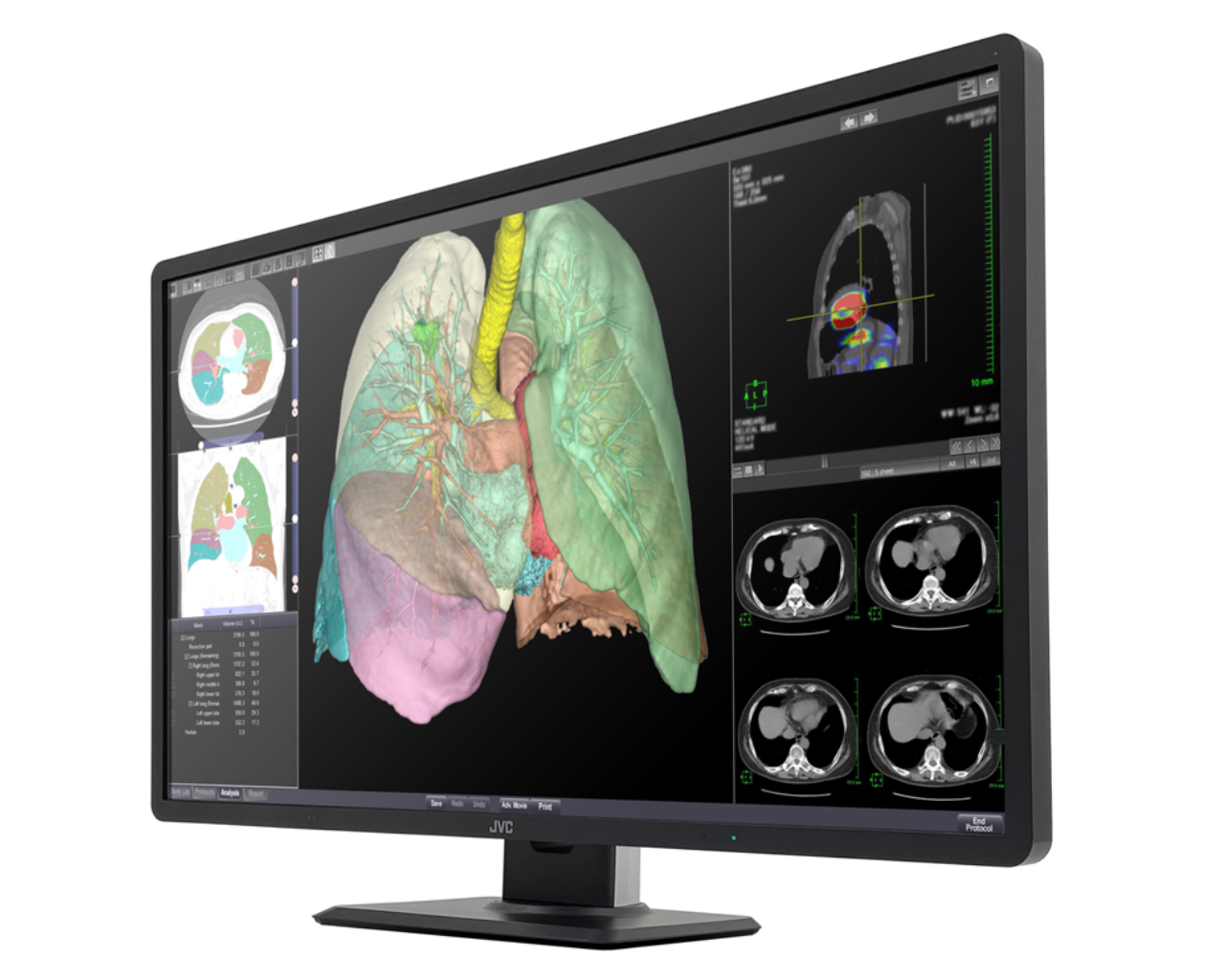
Therefore, we will release the 32-inch wide color monitor which is the largest size of our medical image monitor, “CL-R813” with a high resolution of 8 megapixels (3840 x 2160 pixels). It enables simultaneous display of 6 high-resolution CT / X-ray fluoroscopic images on one screen of this unit, and free layout of various application screens such as viewers, reports, and feedback of AI. In addition, by adopting a narrow bezel and a lightweight design, a slim design is compatible with a large 32-inch screen. Image display, which used to require multiple medical monitors, can now be performed with a single unit, contributing to increasing the work efficiency, reducing the fatigue on doctors who need to read numerous diagnostic images, and saving working space.
Key features
1. 32-inch 8-megapixel monitor for multi-modality environment
The seamless screen with a wide area of 32 inches and a high resolution of 8 megapixels reduces the load of eye movement and provides a comfortable diagnostic imaging environment. Various images such as DR, CT, MRI and pathology can be displayed and it is free to arrange the layout of windows, significantly improving image reading efficiency.
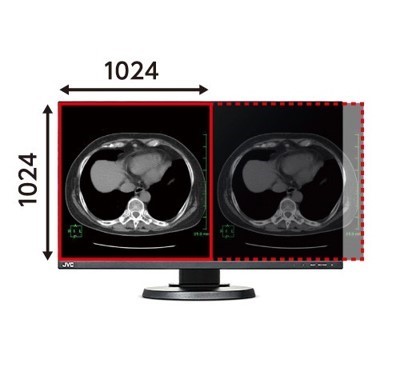
24” FHD Monitor cannot display two CT images on one screen.

CL-R813 can display six CT images on one screen.
2. Narrow bezel and lightweight for slim design
A large 32-inch screen with a narrow bezel and a slim stand footprint realize the lightweight design and make effective use of the working space on the desk.

3. Integrated QA Solution
In order to maintain and manage the display quality of each monitor, QA Medivisor Agent can centrally perform calibration / acceptance test / constancy test / DICOM conformity test complied with guidelines of each countries.
Friendly user interface is adopted for simple operation. Various functions are equipped to reduce the burden of maintenance, such as task scheduling that can automatically execute.
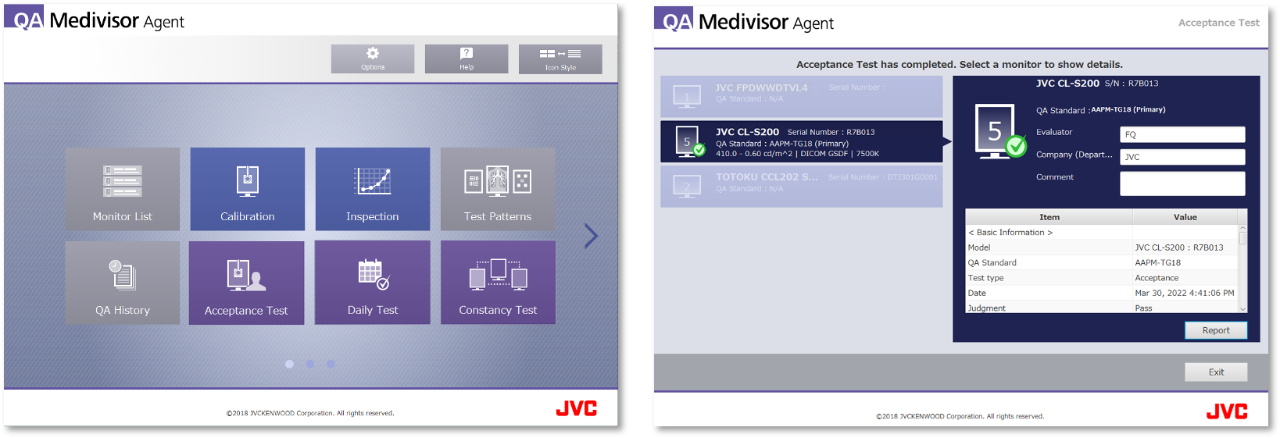
* Optional calibration kit CAL-016 is required for standard QA testing.
4. Built-in Sensor
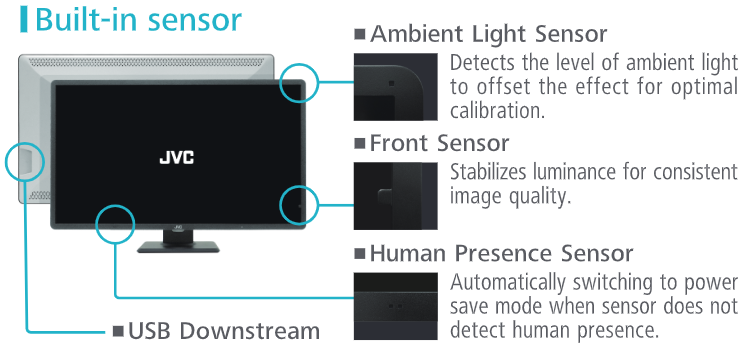
5. Wire Management / DisplayPort Daisy Chains
With the DisplayPort 1.2, the use of daisy chains allows for easier wiring connections.

Specifications
| Model Name | CL-R813 | |
|---|---|---|
| LCD Panel | Technology | 32” color TFT IPS technology |
| Display Area | 708.48 mm × 398.52 mm | |
| Pixel Pitch | 0.1845 mm × 0.1845 mm | |
| Maximum Luminance | 500 cd/m² (typ.) 280 cd/m² (calibrated) |
|
| Contrast Ratio | 1000 : 1 (typ.) | |
| Viewing Angle | 178° vertical and horizontal | |
| Visual Performance | Native Resolution | 3840 × 2160 |
| Display Colors | 16.77 million colors 1.07 billion colors with DisplayPort and 10-bit viewer |
|
| Interface | Input Signal | DVI-D x 1 DisplayPort (DisplayPort 1.2) x 2 |
| Output Signal | DisplayPort (DisplayPort 1.2) x 1 | |
| Plug and Play | DDC2B compliant | |
| Input Power Supply | 100V - 240 V 50/60 Hz | |
| Features | Calibration Control | Luminance, Gamma, Color temperature Capable of storing 3 sets of LUT (Optional Calibration Kit is required) |
| USB Hub | USB Rev.2 compliant, Self-powered USB upstream connector (x1), USB downstream connector (x3) |
|
| Approvals | ANSI/AAMI ES60601-1 (2005) + A1 (2012), CAN/CSA-C22.2 No. 60601-1 (2014), CE (EN60601-1, EN60601-1-2), FCC Part15 subpart B Class B, ICES-003-B, VCCI-B, FDA510(k), J-Moss, RoHS, RCM |
|
| Physical Characteristics | Dimensions(W x H x D) | 742 mm × 487.5~582.5 mm × 196.5 mm |
| Weight | approx. 10 kg | |
| Tilt Stand | Tilt, Swivel | |
| Mount | VESA standard (100 mm x 100 mm) | |
| Accessories | Power cord, DVI cable, DisplayPort cable, USB cable, Operation manual, Installation manual, Software (QA Medivisor Agent LE) | |
<Trademarks>
- QA Medivisor Agent are trademarks or registered trademarks of JVCKENWOOD Corporation.
- All company names and product names contained in this press release are trademarks or registered trademarks of their respective holders.
<「CL-R813」>
Media Contact:
Healthcare Business Division
JVCKENWOOD Corporation
E-mail: medical-display.j@jvckenwood.com
This document is based on the information available to the Company at the time of release and may differ from the latest information.